Originally published by our sister publication, Gastroenterology & Endoscopy News.
The FDA has granted 510(k) clearance to SKOUT (Iterative Scopes), a real-time computer-aided polyp detection device, for adults undergoing colorectal cancer screening or surveillance.
The device’s interface integrates with existing clinical workflows and does not increase total procedure or withdrawal time, according to a press release from the company.
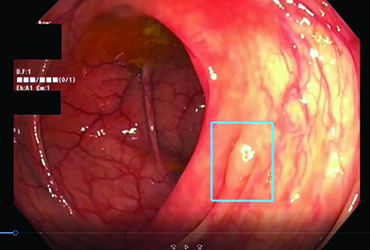
The SKOUT computer-aided polyp detection (CADe) device was evaluated in the largest U.S.-based multicenter study for a CADe to date (Gastroenterology 2022;163[3]:732-741. doi:10.1053/j.gastro.2022.05.028). The investigators prospectively evaluated the use of the CADe device at five academic and community centers by 22 board-certified gastroenterologists. Between January and September 2021, the study enrolled 1,440 patients 40 years of age or older, who were scheduled for screening or surveillance colonoscopy at least three years since their prior examination. Some patients were excluded for incomplete procedure, diagnostic indication, inflammatory bowel disease and familial adenomatous polyposis. The researchers used the computer-generated, random-block method to assign the patients by endoscopist to either the standard (n=677) or CADe colonoscopy (n=682) arm.
The study’s two primary end points were 1) adenomas per colonoscopy (APC), the total number of resected adenomas divided by the total number of colonoscopies, and 2) the true histology rate (THR). The THR is calculated by the proportion of resections with clinically significant histology divided by the total number of resected polyps.
The investigators found that APC increased significantly, by 27%, with use of the SKOUT CADe device, with an average of one additional adenoma resected for every 4.5 patients examined. The investigators found a total of 562 adenomas in the standard arm and 719 in the CADe group. The APC was 0.83 in the standard arm versus 1.05 in the CADe arm (P=0.002).
The investigators also found no decrease in THR when using the SKOUT CADe device (71.7% with standard vs. 67.4% with CADe; P<0.001 for noninferiority).
The SKOUT CADe device demonstrated a 44% relative increase in detection of polyps 5 to 9 mm in size in the proximal colon and a 29% relative increase in detecting these polyps overall. The device also had a higher adenoma detection rate (ADR) than the standard approach, at 47.8% and 43.9%, respectively, but the difference was not significant (P=0.065).
Although ADR is the most recognized indicator of quality colonoscopy, APC is emerging as a significant quality indicator. Aasma Shaukat, MD, MPH, the Robert M. and Mary H. Glickman Professor of Medicine and Gastroenterology at NYU Grossman School of Medicine, in New York City, who has been involved with studies using the CADe device, noted one pitfall of ADR: It can lead to a “one-and-done phenomenon, where the physician doesn’t get credit for finding the extra adenomas” in a given patient.
“Knowing that patients can walk away from their appointment with the confidence that they received a high-quality and detailed colonoscopy is what excites us most about SKOUT’s FDA clearance,” Dr. Shaukat said regarding the recent announcement from the FDA. “Confidence and peace of mind [for] providers and patients alike will allow clinicians to provide stronger recommendations and solutions for their patients.”
—Landon Gray
Dr. Shaukat reported financial relationships with Freenome, Medtronic and Motus GI.


Please log in to post a comment