
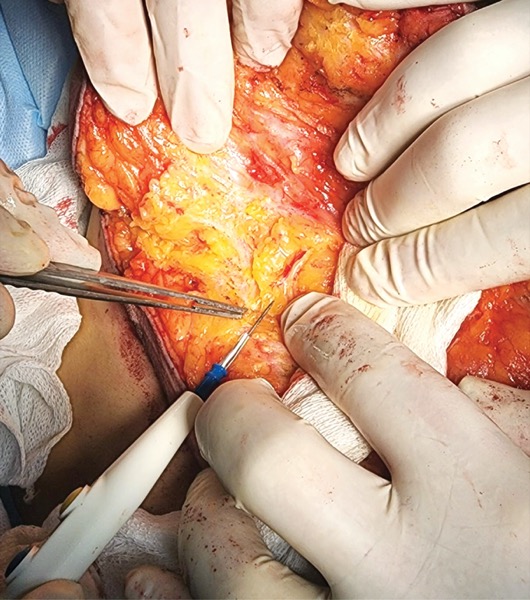
CORAL GABLES, Fla.—Preoperative use of fluorescence in patients undergoing mastectomy can help surgeons identify and map out critical vessels and nerves, not only reducing the risk for the dreaded complication of ischemia and necrosis of the nipple areolar complex (NAC) and mastectomy flaps, but also preserving sensation in the breast.
“Loss of sensitivity is feared by patients, and we have always told them that it’s a largely inevitable effect of mastectomy—after the operation, you will have no more sensitivity of the breast. But now we know this can be modified, and I think, in the near future, it will be possible to have this technology in every operating room,” said Alberto Rancati, MD, the chief of oncoplastic surgery at the University Hospital JosÉ de San MartÍn, in Buenos Aires, Argentina. “It’s amazing. Sometimes I can’t sleep at night thinking about this potential.”
At the 2023 annual meeting of the International Society for Fluorescence Guided Surgery, Dr. Rancati began his talk on FGS in nipple-sparing mastectomy (NSM) with an anatomy review. “There is not only one way to do NSM. Decisions on how to perform the procedure must be made on an anatomical basis for each patient,” he said. “If you can visualize the anatomy, you can place the correct incision, make a safe tumor resection and diminish your risk of complications.”
Ensuring adequate vascularization to the NAC requires careful management of perforator vessels that deliver blood to the remaining flaps. “In mastectomy, you must resect the gland and sacrifice some perforators,” he said, noting that some of them, such as the fifth anterior intercostal artery perforator and the second intercostal perforator vessels, are especially critical.
“If you do a conservative mastectomy and over-dissect the flaps beyond the breast footprint, you can unnecessarily sacrifice the second intercostal artery and the fifth anterior intercostal artery perforator (AICAP) vessels at the inframammary fold, which are important to the nutrition of the NAC after NSM,” Dr. Rancati said in an interview with General Surgery News.
Using fluorescence preoperatively, he and his colleagues have been able to check all of the perforators, some of which may have been interrupted by scars from a previous biopsy or prior radiotherapy, and identify those that will or will not nourish the NAC.
“After doing studies on fluorescence, we decided to move our incision away from the traditional medial site at the intramammary fold—which is exactly where the AICAP vessels are—to the lateral part in order to preserve the AICAP vessels and the second intercostal vessels,” he said.
This change in the team’s approach to mastectomy dramatically diminished their complication rate in terms of ischemia and necrosis. But they also noticed that patients were reporting greater sensitivity in the postoperative breast.
“We were surprised by this, so we kept doing research. Now we have a prototype for a camera that works with a wavelength near fluorescent light,” Dr. Rancati said.
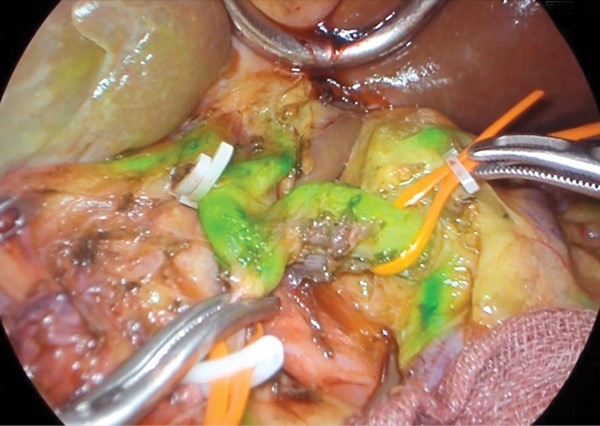
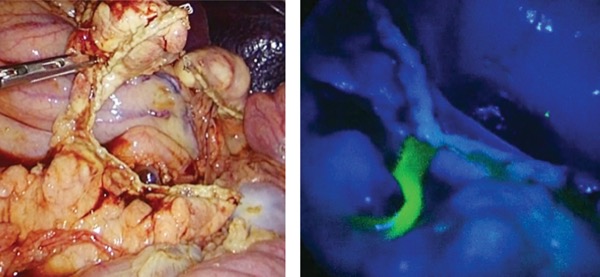
Without this camera, nerves are very difficult to see in the surgical field—unlike vessels, they don’t have a color. “They look like fat. But when you work with this camera, they auto-fluoresce. You see the glowing nerves in the tissue and they are easy to preserve,” Dr. Rancati said, crediting the discovery of this phemonenon to Raul Rosenthal, MD, and Fernando Dip, MD.
“Up to now, with microsurgery, we could graft a nerve taken from another part of the body and recover some sensation. But it makes no sense to sacrifice something and then repair it if you can see the nerve and preserve it,” he noted.
Dr. Rancati and his colleagues published a case report documenting preservation of sensitivity after NSM by preserving the AICAP neurovascular pedicle by moving the incision laterally to a site where it would not cut important nerves. “This is the first case where we preserve sensitivity after NSM, and it was thanks to fluorescence,” Dr. Rancati said (Plast Reconstr Surg Glob Open 2023;11[6]:e5048).
To date, they have operated on 35 patients, preserving the AICAP vessels as well as the second intercostal and lateral thoracic vessels, along with nerves.
“I think we are changing the era, making procedural decisions based on imaging. Because using fluorescence today, it’s like doing surgery with a GPS. You know exactly where you are cutting, what you are doing, making surgery faster and safer. Also, it’s fun for us to know what we are doing and to see it,” Dr. Rancati said.
Innovation for Fluorescence Imaging in Hepatopancreatic Biliary Surgery
CORAL GABLES, Fla.—Fluorescence imaging can be used in practically all surgery, “literally from brain to toe,” said Takeaki Ishizawa, MD, PhD, speaking at the 2023 annual meeting of the International Society for Fluorescence Guided Surgery.
For hepatopancreatic biliary (HPB) surgery, researchers are exploring ways to use fluorescence for perfusion assessment, visual assessment of pancreatic tumors and sentinel node mapping—“though this is the concept of surgery, not the standard,” Dr. Ishizawa, a professor of hepatobiliary pancreatic surgery at Osaka Metropolitan University, in Japan, told General Surgery News.
Perfusion assessment, to confirm arterial blood flow to the liver before dividing the gastroduodenal artery, is fairly straightforward, he said. “We just need a bolus injection of 2.5 mg of indocyanine green (ICG).”
This approach to perfusion assessment can be especially useful in some of the more complex HPB operations, such as Whipple procedures with portal vein resections and distal pancreatectomy with celiac axis resection, confirming perfusion after anastomoses in the former, and visualizing perfusion to the liver and stomach before dividing arteries in the latter, Dr. Ishizawa said.
The use of fluorescence imaging for sentinel lymph node mapping in pancreatic surgery, however, is in a more conceptual phase. A 2018 study showing that the use of ICG–near-infrared imaging detected less than 70% of both regional lymph nodes and para-aortic lymph nodes indicated the procedure needed improvement (Photodiagnosis Photodyn Ther 2018;24:274-279).
“Some surgeons have tried to use fluorescence imaging to visualize the lymphatic flow in pancreatic surgery to minimize the area of lymph node dissection, but I don’t think this is standard procedure,” Dr. Ishizawa said.
Likewise, when it comes to identifying pancreatic neoplasms, while fluorescence imaging with ICG is becoming more widely accepted for liver cancer, it’s not quite where it needs to be to become standard for pancreatic cancer surgery.
“In the field of pancreatic cancer, the application is still very rare,” Dr. Ishizawa said. “Some have reported use of ICG fluorescence imaging for detection of pancreatic tumors, but the detection rate and tumor-to-background contrast is insufficient. Basically, we need new techniques for fluorescence imaging to be used in pancreatic surgery for the detection of tumors.”
To this end, researchers and surgeons have been developing new, cancer-specific fluorophores. On the cutting edge is a carcinoembryonic antigen–targeting fluorophore developed by Alexander L. Vahrmeijer, MD, PhD, whose early research showed use of this fluorophore is safe and feasible (Ann Surg Oncol 2018;25[11]:3350-3357). “They are now conducting phase 2 and higher-phase clinical trials for this probe to be delivered into the OR,” Dr. Ishizawa said.
In their own research, Dr. Ishizawa and his colleagues have developed a probe that targets pancreatic juice, which is clear and practically invisible during surgery, and can cause massive postsurgical bleeding if undetected (Br J Surg 2013;100[9]:1220-1228). “All surgeons want to prevent a pancreatic leak. When this probe, activated by pancreatic chymotrypsin, is sprayed onto the filter paper that was attached onto the pancreatic stump during surgery, and fluorescence imaging is performed, we can see the pancreatic juice leakage,” he said.
Using fluorescence imaging to investigate the causes of pancreatic leakage in a pig model, he found pancreatic juice leaking from the small holes made by the stapler used to divide the pancreas (Gastroenterology 2015;149[6]:1334-1336). This led Dr. Ishizawa to develop a novel device, the PancLoop, consisting of four needles and three suture loops, that can tighten the pancreatic stump and minimize the small pancreatic duct (Glob Health Med 2022;4[4]:225-229). “I expect that use of this device can reduce the risk for postoperative pancreatic fistulas,” he said.
Dr. Ishizawa envisions a future in which fluorescence imaging will visualize the location of pancreatic tumors more accurately as well as pinpoint sites of pancreatic juice leakage, thus reducing the risk for postoperative complications and improving long-term outcomes of cancer surgery. He also thinks fluorophores might move beyond their usefulness as diagnostic tools to become therapeutic agents.
“Already, in animal care, an ICG compound can be used for photodynamic treatment in cats with facial cancer, followed by photoradiation, to treat without any surgical incision. I think in the near future, fluorescence will be used not just for intraoperative diagnosis, but also for active treatment,” Dr. Ishizawa said.
Dr. Ishizawa reported service as a consultant to Sony Corporation and Kono Seisakusho Co., Ltd.
This article is from the November 2023 print issue.




Please log in to post a comment