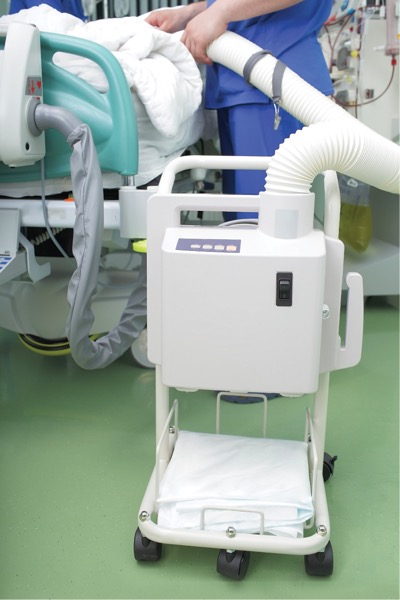
Prewarming a patient prior to surgery has been shown to be important, but using a forced-air warming (FAW) system increases the likelihood of a surgical site infection, according to a recent study.
The retrospective study, which analyzed data from the CDC’s healthcare-associated infection prevalence 2018 survey, found that of 320 pathogen isolate samples collected in the OR of procedures employing an FAW system, 42.5% were higher than the minimally accepted pathogen levels (Ann Med Surg [Lond] 2022;73:102976).
Overall, 3.4% of OB/GYN cases, 5.6% of colon cases, 1.4% of gastrointestinal cases and 5.3% of amputation cases developed an ssI.
“I am always eager to identify any risk-prone areas in healthcare,” said lead investigator Victor Lange, PhD, JD, a hospital epidemiologist for AHMC Healthcare, a system of 10 hospitals in California. “Often, risk is low-hanging and can be mitigated easily once it has been identified.”
In this case, identifying the disadvantages of FAW devices has shed new light on infection risk in the OR, according to Dr. Lange. “Though some reviews on the subject are inconclusive, a number of studies point toward a surface component and tissue–air risk connection,” he said. “Therefore, I wanted to prove the correlation between FAW and consequential ssI risk.”
Study data confirmed that FAW systems do indeed contribute to air contaminants in the surgical environment, thereby leading to an increased risk for cross-contamination and infection. “As with many devices that may contain areas that are not easily disinfected, movement of air across a contaminated surface has demonstrated infection-seeding risk,” he said.
Despite increased contamination risk with FAW systems, “keeping a patient warm helps limit the risk for postoperative infections, decreases bleeding and promotes faster recovery,” Dr. Lange said. “If we fail to actively warm the patient, hypothermia may occur, in addition to complications with increased blood loss or even cardiac events.”
Raising awareness that FAW systems lead to increased risk for contamination should encourage surgical departments to review their disinfection protocols and to identify alternative devices for patient warming, such as blankets, fluid-warming devices and conductive-fiber warming blankets.
“Clinicians must understand the various patient-warming techniques, including the risks and benefits of each,” Dr. Lange said. “As the medical field advances with technological improvements, a host of unforeseen risks may be inherent. Ensuring that the technology is clearly evaluated is paramount to the appropriate use of the device for the intended purposes.”
Gregg Lobel, MD, the chief of anesthesiology and critical care at Englewood Hospital, in Englewood, N.J., said the risk for an ssI always exists and that he does not believe the small study size of 320 total samples “can determine that the risk was greater when FAW devices were used to maintain normothermia.”
Because the study was a retrospective review and not a randomized controlled trial, “it is very hard to draw any conclusions,” said Dr. Lobel, who was not involved with the study. “There are many variables that should be in place to prevent ssIs. Without knowing all of them, it is impossible to blame or applaud FAW.”
As an example, were prophylactic antibiotics given at the appropriate time? Did patients have other risk factors for ssI, like diabetes? What prep solutions were used and were they used properly? Was hand hygiene performed appropriately?
“Our goal is always to have zero ssIs, so even one ssI is unacceptable,” Dr. Lobel said. “Although the number of ssIs has decreased over the past decade due to many new protocols put in place, we still have a ways to go.”
This article is from the December 2022 print issue.


Please log in to post a comment
If only it were true that this equipment was associated with a higher SSI rate (which was convincingly reported in the above article and was certainly not proven in the citation (Ann Med Surg [Lond] 2022;73:102976)- to quote "Overall, 3.4% of OB/GYN cases, 5.6% of colon cases, 1.4% of gastrointestinal cases and 5.3% of amputation cases developed an ssI." These are totally acceptable rates of SSIs for these procedures. AND if only the authors (both in the article and citation) had demonstrated some level of concordance between the pathogens/strains present in the OR and those in the SSI AND if only it were true that under today's mandated asepsis protocols (fast tracking ERPs, antibiotics, skin decontamination, etc), most SSI are actually due to an intraoperative contamination event, then maybe the headline, "Forced air-warming system linked to increased risk for SSI" could be more than a headline. A response such as "we can only control what we can measure and control" is not going to lead to "zero SSIs" under the reasoning that "if some is good, more must be better." A more in-depth understanding of why SSIs develop in the first place is now within our reach and complete intraoperative sterility may not only be impractical/impossible, but itself may not be justified.