LAS VEGAS—Ninety days of preoperative endocrine therapy in women undergoing breast-conserving surgery for invasive breast cancer appears to influence patient preferences and physician recommendations for postoperative radiation, according to primary results from the POWER trial.
Long-term data support the omission of radiation therapy in older women with early-stage estrogen receptor (ER)-positive breast cancer who undergo adjuvant endocrine therapy (AET), but a majority of women still undergo radiation, raising concerns about overtreatment, said Shayna Showalter, MD, a professor of surgery and breast surgical oncology at the University of Virginia School of Medicine, in Charlottesville.
“This trend is driven by multiple factors, including poor tolerance to AET, especially among older women, and the lack of individual patient-level tolerance data about adjuvant endocrine therapy. Forgoing radiation therapy has its own risks, particularly if patients fail to adhere to AET, which can result in undertreatment. Thus, there is a need for individualized treatment plans,” she said.
The POWER trial included women 65 years of age and older with ER-positive, progesterone (PR)-positive/PR-negative invasive breast cancers 2 cm or smaller, with clinically node-negative disease. All patients received 90 days of preoperative ET before undergoing lumpectomy, after which patients and their treating physicians made decisions about radiation therapy.
The primary goals of the trial were to assess whether 90 days of preoperative ET (pre-ET) changed patient and/or surgeon preference for adjuvant radiation therapy. For each cohort, a change in preference was tested from an assumed low change rate of 5% to 15% or more with a one-sided 5% level binomial test.
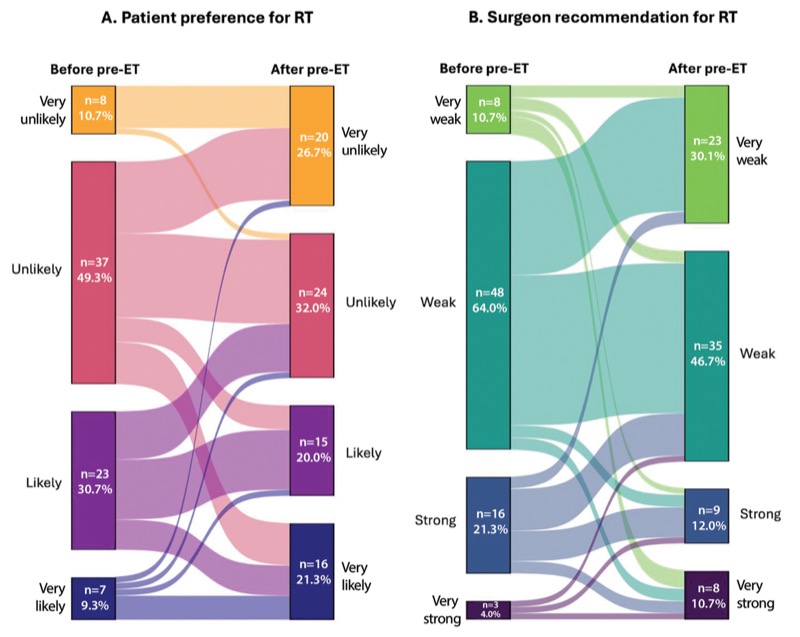
“The strength of radiation preferences and surgeon recommendation for radiation were obtained from the participant and the surgeon at baseline and after the 90-day course of ET. We also obtained preliminary data on the impact of preoperative ET on decisional conflict and decision regret,” Dr. Showalter said.
The trial included 75 women, with a median age of 73 years. Eight participants (10%) stopped using pre-ET early, mainly due to side effects, and 35 (47%) reported adverse events such as fatigue, hot flashes, and arthralgias.
After pre-ET, 21 patients changed their preference for or against radiation, and surgeons changed their recommendation for or against radiation for 18 patients.
“Both of these exceeded the predetermined thresholds constituting a significant change. Interestingly and importantly, the agreement between preferences between patients and surgeons significantly increased, from 53% before pre-ET to 81% after,” Dr. Showalter said.
Overall, patients reported a low level of decisional conflict with no significant change between the time points, and this remained true even in the patients who changed their preference for radiation therapy.
Although not the primary goal of this analysis, patients were categorized at the one-year follow-up based on whether they underwent radiation and whether they initiated and remained adherent to AET.
“The majority of the patients were appropriately treated (surgery and radiation or surgery and AET), while only a minority were undertreated (surgery alone) or overtreated (surgery, radiation, and AET). Adherence to AET remained high at one year, with less drop-off than reported in a historical cohort,” Dr. Showalter said. She hypothesized that this high rate of adherence may be explained by the pre-ET phase, as only patients who tolerated pre-ET went on to initiate AET, making them more likely to remain adherent.
Dr. Showalter concluded that the primary results of the POWER trial support the use of pre-ET as an innovative method to inform adjuvant radiation therapy decisions and can present an opportunity to assess patient tolerance to ET before making adjuvant therapy decisions.
“The new and ongoing POWER II trial is a randomized controlled trial designed to figure out the impact of preoperative ET on over- and undertreatment. This method has the potential to create a paradigm shift in the treatment of older women with early-stage ER-positive breast cancer,” Dr. Showalter said.
Anna Weiss, MD, an associate professor at the University of Rochester Medical Center, in New York, fully endorses pre-ET “to gauge side effects for patients and to inform their future decisions. It’s a smart, easy, and highly informative approach: You give patients the medicine they’ll need anyway to see if they’re going to tolerate it, and if they don’t, you have that information for your radiation consultation, which can really influence what they do.”
Dr. Weiss also noted, “It’s not like we’re pushing patients away from radiation therapy or away from ET, which I think is a concern sometimes with radiation oncologists or medical oncologists—that if you expose patients to ET and they don’t like it, they just won’t take it. Based on the evidence, we’re not really seeing that.”
Rather, what we’re seeing is a more nuanced approach to treatment, she said. “Instead, it’s helping patients make more informed choices, and reassuring radiation oncologists that most patients will adhere to their ET. This allows for more tailored adjuvant treatment.”
This article is from the November 2025 print issue.



Please log in to post a comment