For surgical practitioners, management of intraabdominal infections (IAIs) has grown more challenging in recent years due to the aging population, the growing burden of chronic disease in older people, and the increased prevalence of resistant bacteria and fungi both in the health care setting and in the community. Those trends, along with the advent of newer approaches to source control and the introduction of new antibiotic agents, helped prompt the Surgical Infection Society to update its guidelines on the management of IAIs,1 which were most recently issued in 2010 as a joint guideline with the Infectious Diseases Society of America.2
As director of the task force charged with revising the 2010 guidelines, I sought to facilitate discussion of several key issues pertaining to the treatment of patients with IAIs. A common thread running through these issues was the concept of antimicrobial stewardship, which refers to coordinated interventions to promote appropriate use of antimicrobial agents. Our recommendations thus reflect the task force’s embrace of those principles, which we believe are essential to optimizing outcomes in patients with IAIs.
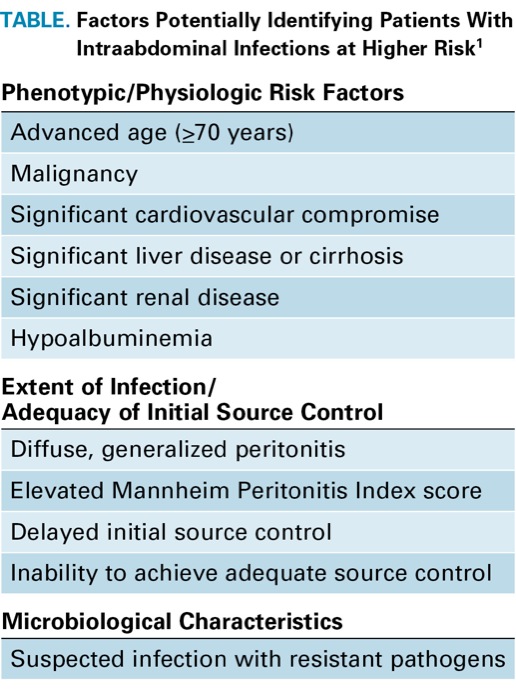
Intrinsic to the concept of antimicrobial stewardship is a systematic approach to risk assessment, a key factor in directing antimicrobial therapy appropriately to the individual patient. A subgroup headed by Addison May, MD, extensively reviewed this topic. Patients meeting the criteria for sepsis or septic shock and those with elevated Acute Physiology and Chronic Health Evaluation II (APACHE II) scores are known to be at high risk. However, many patients with IAIs will not meet the criteria for sepsis or septic shock, and many are not evaluable via APACHE II scores, which can be difficult to calculate in an urgent situation and are subject to inter-rater variability. Recognizing that such patients may have a number of risk factors for an adverse outcome, the revised guidelines suggest that patients having two or more specific risk factors should be considered higher risk (Table). At the same time, whereas the guidance to direct narrower-spectrum therapy to patients at lower risk has appeared in previous iterations of the SIS guidelines, the revised version makes this more explicit, as the increased prevalence of resistant bacteria has made it especially important to identify potential candidates for less aggressive and shorter-duration antimicrobial therapy.
| Table. Factors Potentially Identifying Patients With Intraabdominal Infections at Higher Risk1 |
| Phenotypic/Physiologic Risk Factors |
|---|
| Advanced age (≥70 years) |
| Malignancy |
| Significant cardiovascular compromise |
| Significant liver disease or cirrhosis |
| Significant renal disease |
| Hypoalbuminemia |
| Extent of Infection/ Adequacy of Initial Source Control |
| Diffuse, generalized peritonitis |
| Elevated Mannheim Peritonitis Index score |
| Delayed initial source control |
| Inability to achieve adequate source control |
| Microbiological Characteristics |
| Suspected infection with resistant pathogens |
The importance of risk assessment dovetails with source control, another fundamental imperative to antimicrobial stewardship. Good source control obviates the need to use extended-duration antimicrobial therapy in patients with IAIs, and is the leading factor in determining whether patients experience treatment failure or die from these infections. The concept of source control has undergone numerous adaptations as surgical techniques have evolved. Consequently, the revised guidelines emphasize the importance and timing of source control, and are informed by two approaches to source control that have been adjusted over the past five to 10 years. One approach is based on the recognition that minimally invasive surgical procedures are now clearly established, acceptable and preferable in many cases, and that they generally satisfy the tenets of source control. The other approach focuses on criteria for tailoring the procedure according to the disease at hand (i.e., localized vs. diffuse) and the patient’s underlying condition (e.g., sepsis, critical illness). For example, when treating an unstable sick patient, the latter approach may call for a “damage control” laparotomy with a plan of returning and reoperating on the patient later.
The task force also addressed the growing threat of resistant bacteria, and aimed to provide guidance as to when to use supplemental antibiotics in patients with hospital-acquired IAIs in whom one might suspect the presence of resistant or difficult-to-treat organisms, such as Enterococcus, methicillin-resistant Staphylococcus aureus, Pseudomonas aeruginosa, extended-spectrum beta-lactamase (ESBL)- or carbapenemase-producing Enterobacteriaceae, or certain Candida species. We also discussed the major global uptick in resistance to fluoroquinolones, which is not necessarily reflected in increased treatment failure rates in patients with IAIs. Although fluoroquinolones are still listed as recommended agents, the revised guidelines suggest that these agents should be reserved for patients who have major allergic or anaphylactic reactions to beta-lactam antibiotics such as penicillins, cephalosporins or carbapenems. The revised guidelines also consider the effect of resistance on selection of antibiotics in certain regions; a prominent example is the increased prevalence of ESBL-producing Escherichia coli in Asia and Latin America. For those areas, the guidelines recommend considering carbapenems as up-front therapy if there is high-level resistance in the community to common cephalosporins.
Since the 2010 guidelines were issued, two new cephalosporin-beta-lactamase inhibitor combinations, ceftolozane-tazobactam (Zerbaxa, Merck) and ceftazidime-avibactam (Avycaz, Allergan), have been approved. The current guidelines consider use of ceftolozane-tazobactam plus metronidazole as an option for empirical therapy of adults (Grade 2-A recommendation), but reserve this regimen primarily for high-risk patients strongly suspected of being or proven to be infected with resistant strains of P. aeruginosa, for which other agents are not suitable (Grade 2-C). Similarly, the current guidelines recommend the use of ceftazidime-avibactam plus metronidazole as an option for empirical therapy of adults (Grade 2-A), but reserve this regimen primarily for high-risk patients strongly suspected of being or proven to be infected with Klebsiella pneumoniae carbapenemase–producing Enterobacteriaceae, for which other agents are not suitable (Grade 2-C).1
Given the paucity of data pertaining to managing patients who fail to respond to initial therapy, we relied on the expert opinions of the task force members, particularly Dr. May, who championed this section of the revised guidelines. This section emphasizes the importance of differentiating early from late treatment failure, and of tailoring management approaches accordingly. For example, a patient who is still “going downhill” at 48 hours post-surgery does not necessarily reflect a failure of antimicrobial therapy; rather, this situation may signal something gone awry with source control, and should trigger re-exploration of potential early sources of IAIs.
By contrast, for the postsurgical patient whose condition improves over five to seven days but then appears to worsen, the antimicrobial therapy has, in all likelihood, already selected resistant organisms. Imaging studies, followed by percutaneous drainage or other minimally invasive procedures, with antimicrobial therapy directed against potentially resistant organisms may therefore be appropriate for the so-called late-failure patient.
In the absence of culture data, simply adding more or different antibiotic agents to the patient’s regimen may not help, and violates the principles of good antimicrobial stewardship. Oftentimes, the culture data will suggest that the patient can be treated with an agent of a narrower spectrum, possibly bolstering the case for de-escalating antimicrobial therapy. This is not a new concept, but one that is emphasized throughout the revised guidelines.
The task force also considered the duration of antibiotic therapy, noting that the vast majority of patients with IAIs can be managed with shorter courses of therapy, as demonstrated in Sawyer and colleagues’ New England Journal of Medicine seminal publication.3 In this large, prospective randomized trial, Sawyer and colleagues found that four days of fixed-duration antibiotic therapy was more than adequate in the patients who were tested, with outcomes similar to those after a longer course (approximately eight days) of therapy. The revised guidelines therefore position four days as the standard by which duration of therapy should be measured, and there is no reason for a longer course of treatment in the absence of treatment failure or a need for persistent antimicrobial therapy. Indeed, other investigators have followed groups of IAI patients with sepsis or other high-risk factors, and none of these groups have appeared to benefit from a longer duration of therapy.4-6
The revised guidelines also include additional recommendations pertaining to the treatment of pediatric patients with IAIs, developed by a subgroup headed by Evan Nadler, MD. Although there are far fewer data on pediatric patients than adults, our recommendations for managing pediatric patients essentially mirror those we have issued for adults. One nuance is that we recommend limiting antimicrobial therapy to five days (120 hours) in pediatric patients older than 1 month of age who have had adequate source control. Otherwise, the guidelines recommend treating pediatric patients in an analogous fashion to adult patients.
The revised guidelines aim to elevate the principles of antimicrobial stewardship so they are part of routine clinical practice in the management of IAIs in the surgical setting. I invite you to explore the guidelines in detail, and to contact me and the other task force members with your questions and comments.
References
- Mazuski JE, Tessier JM, May AK, et al. The Surgical Infection Society revised guidelines on the management of intra-abdominal infection. Surg Infect (Larchmt). 2017;18:1-76.
- Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Surg Infect (Larchmt). 2010;11:79-109.
- Sawyer RG, Claridge JA, Nathens AB, et al. Trial of short-course antimicrobial therapy for intraabdominal infection. N Engl J Med. 2015;372:1996-2005.
- Rattan R, Allen CJ, Sawyer RG, et al. Patients with risk factors for complications do not require longer antimicrobial therapy for completed intra-abdominal infection. Am Surg. 2016;82:860-866.
- Rattan R, Allen CJ, Sawyer RG, et al. Patients with complicated intra-abdominal infection presenting with sepsis do not require longer duration of antimicrobial therapy. J Am Coll Surg. 2016;222:440-446.
- Hassinger TE, Guidry CA, Rotstein OD, et al. Longer-duration antimicrobial therapy does not prevent treatment failure in high-risk patients with complicated intra-abdominal infections. Surg Infect. 2017;18:659-663.
Dr. Mazuski is professor of surgery, Division of General Surgery, Acute and Critical Care Surgery Section, Washington University School of Medicine in St. Louis.


Please log in to post a comment