In 1996, I attended my first bariatric surgical conference: the 13th annual meeting of the American Society for Bariatric Surgery (now the American Society for Metabolic and Bariatric Surgery) at the ChÂteau Frontenac, in Quebec City. It was a modest gathering. Instead of a bustling convention center with rows of vendors, there were perhaps six exhibitors with amateur displays set up in a cramped side room. Bariatric surgery was then a fringe subspecialty of general surgery, fighting for legitimacy. Few academic centers had substantial interest in the subspecialty, and the majority of cases were performed in smaller community hospitals—many with, at best, mixed reputations. Unlike transplantation, cardiac, or oncologic surgery, bariatrics had few marquee names. The surgical icons of the era—Thomas Starzl, Michael DeBakey, Denton Cooley, David Sabiston, Frank Spencer, John Cameron, Norman Shumway—were not bariatric surgeons.
The Early Debates
Even in those early years, the same arguments we still hear today were already being voiced. Proponents of Dr. Edward Mason’s vertical banded gastroplasty warned of anemia and bone loss with gastric bypass. Advocates of gastric bypass, stimulated by Dr. Harvey Sugarman’s work, countered that restrictive procedures failed for “sweet eaters” and grazers. Dr. Nicola Scopinaro argued both sides were wrong—only a distal bypass could ensure durable results. His view was echoed by Drs. Picard Marceau and Douglas Hess, who promoted the duodenal switch, preserving the pylorus with a longitudinal gastric resection.
Of greater historical consequence was the arrival of laparoscopy. Drs. Wesley Clark and Alan Wittgrove stunned the audience with their initial results of laparoscopic gastric bypass. Dr. Robert Greenberg reported on laparoscopic adjustable banding at Mount Sinai. The excitement was amplified by Dr. Walter Pories’ landmark 1995 Annals of Surgery (222[3]:339-350) paper documenting remission—and in some cases, cure—of type 2 diabetes after gastric bypass. For the first time, bariatric surgery seemed poised to enter the mainstream.
Rapid Expansion
By 2001, the ASMBS meeting in Washington, D.C., drew 1,700 surgeons and a convention hall-packed with exhibitors. The small community hospital origins of the field gave way to presentations from major academic centers. Institutions were racing to recruit the limited number of surgeons trained in advanced minimally invasive techniques.
Industry accelerated the growth. Companies developed staplers, energy devices, and instrumentation tailored to laparoscopy. Endoscopic staplers and vessel-sealing tools made these operations safe and reproducible. Hospitals and device makers alike recognized that bariatric surgery wasn’t just a conversion from open procedures—it represented a new patient population. With obesity rates climbing, bariatric surgery was an untapped market that promised profitability, media attention, and prestige. Celebrities such as Al Roker, Carnie Wilson, and Star Jones brought further public visibility, even glamor.
Technical mastery of complex laparoscopic reconstructions created a new class of highly regarded surgeons. Residents competed fiercely for fellowships. By 2010, annual U.S. volume had grown from less than 10,000 cases in 1990 to more than 200,000.
Yet, despite this growth, penetration remained minimal. Perhaps 50 million Americans could benefit, yet fewer than 250,000 procedures were being performed each year. Remarkably, that number has been essentially stagnant for the past 15 years.
The GLP-1 Era
For decades, people speculated that surgery would disappear once a “magic pill” was discovered. That pill, many believe, arrived with the glucagon-like peptide-1 (GLP-1) receptor agonist. Exenatide in 2005 and liraglutide in 2010 demonstrated weight-loss potential. Semaglutide (Ozempic, Novo Nordisk), approved in 2017, seemed transformative: The SUSTAIN-6 trial showed cardiovascular benefit in diabetes (N Engl J Med 2016;375[19]:1834-1844); SUSTAIN-7 reported 12% to 15% average body weight loss (Lancet Diabetes Endocrinol 2018;6[4]:275-286]; and SURPASS for tirzepatide (Mounjaro, Eli Lilly) was even more impressive (N Engl J Med 2021;385[6]:503-515). Public perception declared the revolution complete. Bariatric surgical volumes fell by 30% to 40% in the last several years.
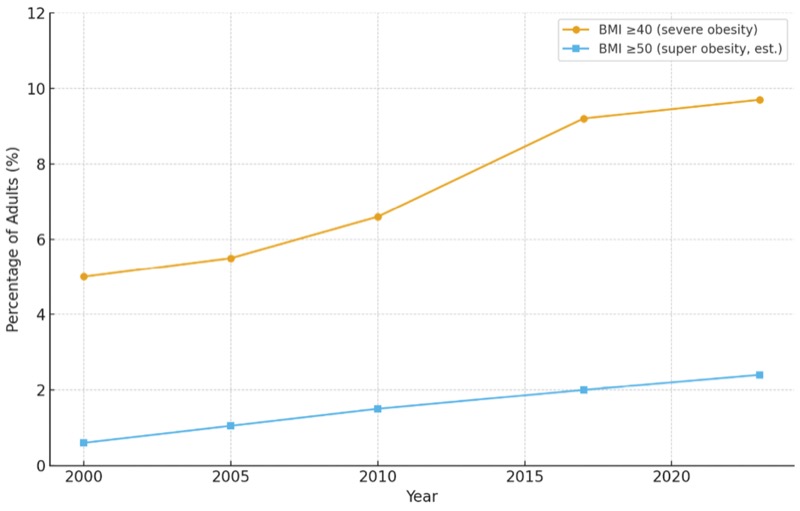
But reality is more sobering. In 2017, there were 462 million people worldwide with type 2 diabetes. Today, according to the International Diabetes Federation, that number is 550 million In the United States, the prevalence of people with a body mass index of at least 40 kg/m2 has risen from 9.2% in 2017 to higher than 10% today, with a BMI of at least 50 kg/m2 approaching 2% and steadily increasing. GLP-1 therapies may have expanded the pool of weight-conscious patients, but they have not reversed the epidemic.
Gender Disparities
Treatment utilization is also skewed. Women account for 68% of all Ozempic prescriptions and more than 80% of those for weight-loss indications (Wegovy [semaglutide], Novo Nordisk; Zepbound [tirzepatide], Lilly). Bariatric surgery shows the same bias: 75% to 85% of patients are women. Meanwhile, men—who already have shorter life expectancy—are undertreated despite higher obesity-related risk.
Survival, Not Preference
Analysis of Northwell’s database of 570,000 adults illustrates the stakes. The prevalence of a BMI of at least 40 or at least 50 kg/m2 rises with age until the fifth decade of life—then declines rapidly. Simply put, men with a BMI more than 50 kg/m2 rarely reach the age of 60. In women, the noticeable decline in prevalence is a decade later. These patients aren’t “losing weight with age.” They are dying prematurely.
The story of Anita Feinberg, one of the first duodenal switch patients at Lenox Hill in 2001, underscores this. With a BMI greater than 70 kg/m2, she faced disability and shortened survival. Now 74, she exercises daily and enjoys time with her grandchildren. It is inconceivable that she would be alive and thriving without surgery. Yet rarely is this information objectively conveyed to individual patients. They may be told to lose weight or asked whether they are interested in pursuing weight-loss alternatives. But, the dangers of their obesity are not objectively measured, nor the risks explained. Furthermore, often they are preoccupied with their comorbid conditions, and less focused on the underlying cause.
Why the Stagnation?
Obesity is treated as a lifestyle issue or risk factor, not as a disease that directly causes death and disability. Heart disease and cancer prompt staging, multidisciplinary review, and aggressive intervention. Severe obesity—an equally lethal condition—does not. Hospital admissions for cellulitis, chronic obstructive pulmonary disease, heart failure, stroke, or cancer are often driven by obesity, yet the connection is rarely made explicit. As a result, if a person with considerable obesity is admitted for respiratory issues, the diagnosis, reimbursement, and management are based on a diagnosis of exacerbation of a pulmonary condition—not high-stage obesity with respiratory impairment. Both patients’ and physicians’ focus is on the acute issue, and the contribution of the underlying condition is less likely discussed or managed.
Meanwhile, GLP-1 trial results are best-case scenarios. Nearly 20% of patients in trials are nonresponders, despite strict exclusion of those least likely to succeed (very high BMI, weight-gain medications, orthopedic limitations). Recidivism and partial response are common in practice. Drugs will improve, but they will not eliminate the need for surgery.
A Path Forward
The true barrier is the absence of staging. Cancer is treated based on tumor grade and spread, coronary disease on anatomic severity. Obesity care is still driven by patient preference. There is no linear relationship between those whose life expectancy is greatly reduced and those who have surgery. Dr. Arya Sharma’s Edmonton Obesity Staging System is a step in the right direction but has not been widely adopted. Dr. Sharma combines BMI with measures of function to stage obesity. Those with stage 4 disease are close to incapacitated. Those with stage 3 have a markedly reduced life expectancy. If all patients were staged, it would become much more practical to determine the optimal medical pathway. It would also allow for multicenter studies that would drive clinical standards. Cancer patients are not asked what therapy they are interested. They are assessed, staged, and, following complete workup, presented with the best option to maximize lifespan, quality, and morbidity. There will always be patient choice, but it is based on likely outcomes based on their level of disease.
The future of bariatric surgery rests on proving survival benefit and making surgery standard of care for defined subgroups. Men in their 30s with a BMI higher than 50 kg/m2 rarely reach the age of 60. Similarly, women like Anita Feinberg with a BMI of 70 kg/m2 would not be alive or thriving without aggressive intervention. We need to define cohorts where surgery is more than optional, but strongly suggested and lifesaving.
That requires multicenter trials that enroll high-risk patients, quantify their baseline loss of life expectancy, and measure how surgery changes it. End points must go beyond weight loss and comorbidity resolution. They must be survival and disability-free years or their markers. We need to create a culture in which every patient with obesity is staged and in which suggested remedies follow an appropriate disease management strategy based on this information.
Conclusion
For decades, bariatric surgeons focused on quality, technical innovation, and expanding indications to lower BMI groups. Outstanding research showed the value for diabetes, sleep apnea, and cancer and heart disease risk reduction. Unfortunately, to date, bariatric is something to consider but not a mandatory referral for any patient or cohort. The next era must focus instead on disease stratification and documenting which treatments best improve life expectancy. In contrast to cancer, the survival rate for even advanced-stage patients makes it essential that we use proxies for life expectancy.
The evaluation of bariatric patients has to be more comprehensive than BMI and assessment of comorbid conditions. Overall function needs to be measured. By far, the best method is maximal oxygen consumption testing. Those with low values have a two- to four-fold greater risk for mortality. Once it is about survival, the thought process is vastly different. To this point, many patients who had undergone bariatric surgery were at increased risk for chronic disease. Our goal is to identify the patients who we can clearly show have reduced life expectancy, and make surgery standard of care for those cohorts.
Surgery will never be universally popular. But if it is clearly shown to prevent disability and prolong life, it will remain indispensable—even in the Ozempic era.
Dr. Roslin is the Director of Bariatric and Metabolic Surgery at Lenox Hill Hospital, in New York City, and Northern Westchester Hospital, in Mount Kisco. He is a Professor of Surgery at The Barbara and Donald Zucker School of Medicine at Hofstra/Northwell, in Hempstead, New York.
This article is from the November 2025 print issue.



Please log in to post a comment
Mitch thank you! Great vision.
tumor boards have worked well, where cardiac care seems to have stayed in silos
How do we build collaboration that PULLS patients in from PCP's, as well as sleep labs and orthopedic care?