CHICAGO—The development of a novel suture-tension distribution device may spell relief for people who undergo high-risk oncologic laparotomy, according to a pilot study. The investigation found a very low rate of incisional hernia in the first year after such procedures, which the researchers said may have a profound impact on the quality of life of these patients.
“All surgeons know what happens when we have sutures that are very tight and stress the tissue to the point where it eventually tears,” said Juan Camilo Cote Correa, MD, a professor of surgical oncology at the University of Antioquia in Medellin, Colombia. “We’ve tried multiple techniques, but none of them seem to work perfectly. So, what are we to do?”
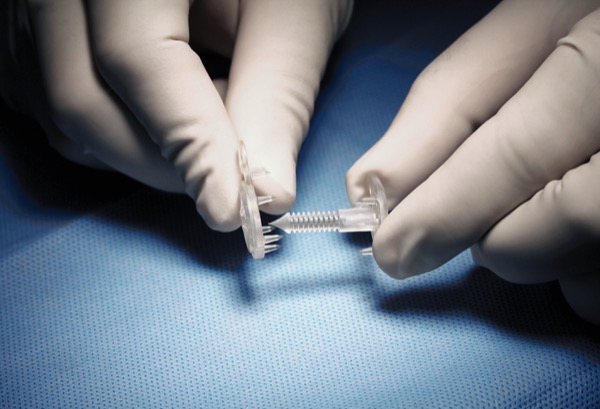
Enter REBUILD, an investigational, bioabsorbable suture anchor designed to broadly distribute suture tension in tissue before being absorbed within one year of implantation.
“We place the sutures within the device’s eyelets,” Dr. Correa explained. “It’s very easy to handle, and it helps us relieve tension on the abdominal wall. You can think of it the same way as a shoe. If the shoe has no eyelets, the laces can tear the shoe up. But when you have eyelets, they protect the shoe. This device does the same thing.”
To test the safety and efficacy of the device, Dr. Correa and his colleagues enrolled 15 adult oncology patients (median age, 56 years; 10 males) scheduled for elective laparotomy into a prospective, multicenter, single-arm, first-in-human investigational study. After the oncologic procedure, anchors were placed along the length of the incision and the abdominal wall closed by running a suture through the eyelets of opposing anchors. Abdominal exams were performed at various time points to assess the integrity of the abdominal wall.
The study’s primary end point was a clinically intact abdominal wall at three, six and 12 months. Secondary end points included radiological examination of the abdominal wall, pain, quality of life and device-related adverse events.
Presenting at the 2024 annual meeting of the American Hernia Society, Dr. Correa noted that at 12 months after surgery, 14 of the 15 participants had no clinical evidence of hernia.
“That’s a 7% clinical hernia rate in a population where you would consider a rate of 41% to be normal,” Dr. Correa said.
CT and MRI scans in the 12 patients who completed the study demonstrated no evidence of midline attenuation or hernia in 11 (92%).
“In other words,” he added, “the radiographic hernia rate was only 8%.” The inter-rectus distance of imaged, intact participants ranged from 0 to 19 mm, although seven patients had an inter-rectus distance of 0 mm. “We had great response; it was amazing,” Dr. Correa noted.
With respect to pain, average visual analog scale scores were 1.3 at discharge, 1.2 at three months, 0.3 at six months and 0.1 at 12 months after surgery. Of note, nine of 15 participants were discharged without opioids for pain control. Quality-of-life assessments were commensurate with the low pain scores.
Analyses of safety and tolerability revealed no device-related adverse events, although an adjudication panel determined that the device may have contributed to one incident of skin dehiscence with intact fascia and two superficial wound infections.
“The device tends to be bioabsorbed within a year, and even though you could feel it in some patients, they had no pain whatsoever and no related complications,” Dr. Correa explained.
Session co-moderator Wen Hui Tan, MD, an assistant professor of surgery at the Medical College of Wisconsin, in Milwaukee, found the results encouraging.
“Can you tell us a little more about the device itself in terms of patient selection?” she asked. “Are there patients that would not be the candidates for this?”
“Honestly, I think we pushed the envelope in terms of who got the device,” Dr. Correa replied. “We didn’t cherry-pick our patients; we used it in a high-risk population that we thought would really benefit from the procedure. That said, I don’t really see any contraindications. We didn’t remove it when we had infections, and we didn’t remove it when we had to reoperate. And the benefits that we saw in our cohort were immense.”
The study was sponsored by AbSolutions Med Inc., which manufactures the REBUILD device.