
Welcome to the December issue of The Surgeons’ Lounge. Guest experts Anne Warren Peled, MD, the co-director of the Breast Care Center of Excellence at Sutter Health California Pacific Medical Center, and Ziv M. Peled, MD, of Peled Plastic Surgery, both in San Franscisco, discuss the most common questions in nerve preservation and allografting for sensory innervation following immediate implant-based breast reconstruction. In this issue, we also feature another installment of “The Procedure, the Name,” on the art of fat grafting.

We look forward to our readers’ questions and comments.
Sincerely,
Samuel Szomstein, MD, FACS
Editor, The Surgeons’ Lounge
Szomsts@ccf.org
@YANKEEDOC44
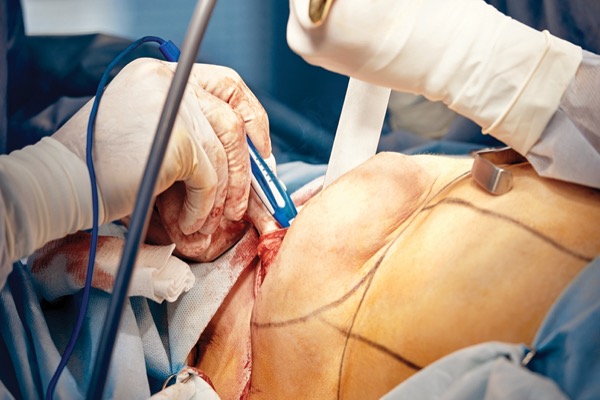
Nerve Preservation and Allografting in Postmastectomy Breast Reconstruction
Anne Warren Peled, MD, and Ziv M. Peled, MD, are interviewed by Lisandro Montorfano, MD, a plastic and reconstructive surgery fellow at Vanderbilt University Medical Center, in Nashville, Tenn.
Dr. Montorfano: What is nerve preservation and allografting for sensory innervation following immediate implant-based breast reconstruction?
Drs. Warren Peled and Peled: Over the past several decades, medicine has made tremendous improvements, not only in the surgical oncologic treatment of breast cancer but also in terms of aesthetic reconstruction. However, one of the remaining hurdles is postoperative sensation, which tends to be extremely poor or completely absent in the vast majority of cases. This problem presented us with an opportunity and spurred us to create an operation to optimize postmastectomy sensation in patients undergoing mastectomy with implant reconstruction. Similar attempts have been made with autologous tissue reconstruction; however, 80% of breast reconstructions performed in the United States annually are implant-based. Our technique involves preserving sensory nerves whenever possible, and reconstructing them when portions of the nerves need to be resected for oncologic reasons.
Dr. Montorfano: Could you describe the technique you use to preserve sensation on this patient population?
Drs. Warren Peled and Peled: The ability to preserve sensation really relies on favorable anatomy. In other words, if the lateral, superficial intercostal nerves (usually T3-T5) happen to travel directly laterally into the skin/subcutaneous tissue flap created during a mastectomy, those nerves can be identified as they emerge from the chest wall and dissected away from the breast tissue. In that way, they can be gently retracted away from the resected tissue, leaving them uninjured from the mastectomy itself and preserved in their entirety without the need for any nerve repair or reconstruction. In the vast majority of these patients, very good sensation returns within a few weeks or months. When the same nerves travel through the breast parenchyma and portions of the nerves need to be extirpated for oncologic reasons, the nerves are reconstructed just as they would in the arm or leg.
Dr. Montorfano: Do you always combine direct-to-implant breast reconstruction with nerve preservation and allografting for sensory innervation?
Drs. Warren Peled and Peled: We offer this technique to every patient who is having an implant-based breast reconstruction procedure. Optimizing postmastectomy sensation has been shown to be associated with improved patient satisfaction and outcomes postoperatively. Doing so also reduces the chances of complications in the distant future, such as wounds stemming from severe abrasions or burns in numb skin, which can have devastating complications for the underlying reconstruction, with exposed or infected implants that then need to be removed and reinserted at a later date. Furthermore, postmastectomy pain syndrome remains a huge problem, with 20% to 70% of patients complaining of chronic pain after their operations. While the etiology of the latter problem is likely multifactorial, it is reasonable to assume that a significant number of those patients have neuropathic pain stemming from transected and non-reconstructed intercostal nerves. We have now performed over 500 mastectomies using this technique and have yet to see a patient with pain issues beyond six months. This indication alone is a reason to consider incorporating such a technique into one’s repertoire.
Dr. Montorfano: Is patient selection important at the time of performing this operation? Is there an ideal candidate?
Drs. Warren Peled and Peled: As with any operation, thoughtful patient selection is critical to achieve optimal outcomes. I certainly think that starting out, the ideal candidate would be a BRCA patient undergoing a nipple-sparing mastectomy who is looking for a modest reconstructive size (B cup or small C cup), for whom a direct-to-implant reconstruction would be possible. Having said that, at this point we have also had success with much larger reconstructions—in cancer patients as well as patients who have had skin-sparing mastectomies—not nipple-sparing mastectomies.
Dr. Montorfano: Can patients expect return of baseline sensation or possibly improved sensation from baseline? What is your experience?
Drs. Warren Peled and Peled: This question really gets at the heart of how one measures sensation. We measure our outcomes in two ways: One is through the use of specific, nipple/areolar-focused, validated questions from the BREAST-Q. At this point, with a minimum of six months’ postoperative follow up, 66% of patients are reporting some or a lot of sensation in their nipple/areolar complexes, and over 90% of patients are reporting some or a lot of overall breast sensation. In addition, over 75% of patients say their nipples are very or somewhat responsive to touch, and nearly 80% of patients are reporting that their overall breast area plays a very important or somewhat important role in intimacy after mastectomy. Second, we use a pressure-specified sensory device that is more accurate and precise when compared with Semmes-Weinstein monofilaments. Using this instrument, we are finding that in all areas tested, with a minimum six-month postoperative follow-up, over 80% of patients have good to excellent sensation. We have also had a few patients who have reported slightly improved sensation postoperatively.
Dr. Montorfano: While preserving nerves, are there any concerns regarding the oncologic outcomes of the operation? How does radiation therapy affect sensation after nerve preservation and allografting surgery?
Drs. Warren Peled and Peled: From an oncologic standpoint, we have only had one recurrence in almost five years of using this technique. While this is not yet long-term oncologic follow-up, we believe that this follow-up is simply a matter of time. Furthermore, it is important to keep in mind that the mastectomy procedure itself remains exactly as it was before we started using this approach. In other words, we do not leave any breast tissue behind when preserving or reconstructing nerves. These structures themselves are only 1.2 mm in diameter on a good day and easily distinguished from breast tissue in experienced hands.
Dr. Montorfano: Is this technique applicable in academic institutions? What advice would you give to academic breast and plastic surgeons?
Drs. Warren Peled and Peled: We believe that this technique is applicable in any practice setting as long as time and patience are given to incorporate it thoughtfully and carefully. We have taught breast surgeon/plastic surgeon duos from both academic institutions as well as private practices; however, this technique requires input from both physicians and requires both to be present in the OR throughout the case, which often requires a shift in practice patterns. Nonetheless, we firmly believe that the ability to regain as much sensation as possible after mastectomy is critical to reduce the long-term sequelae of breast cancer treatment, which is so important given that women are now living many decades with their diagnoses and treatment decisions.
Suggested Reading
- Peled AW, Peled ZM. Nerve preservation and allografting for sensory innervation following immediate implant breast reconstruction. Plast Reconstr Surg Glob Open. 2019;7(7):e2332.
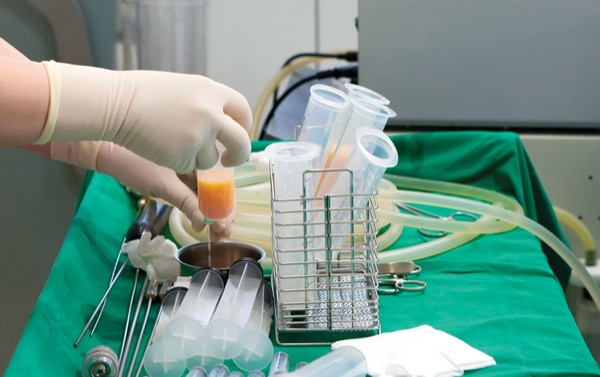
The Procedure, the Name: The Art of Fat Grafting
By Lisandro Montorfano, MD, a plastic and reconstructive surgery fellow at Vanderbilt University Medical Center, in Nashville, Tenn., and Mustafa Tamim Alam Khan, DO, a general surgery resident at The University of Texas Health, in San Antonio
Initial experiences with fat grafting were described by German surgeons between the 19th and 20th centuries. In 1893, Gustav Neuber (1850-1932) provided the first description of fat transplantation during a meeting of the German Society of Surgery. Neuber transferred harvested adipose tissue to the orbital region to correct a scar that had resulted from osteomyelitis. At the time, he recommended that small volumes of fat—no larger than a pea—be injected to allow for neovascularization to occur.1 Two years later and also in Germany, Vincenz Czerny (1842-1916) described the first case of breast reconstruction, following partial mastectomy for fibrocystic disease, using a lipoma from the patient’s flank. The patient tolerated the procedure very well and no adverse reactions were described.2
In 1910, Erich Lexer reported using a block of subcutaneous tissue harvested from the abdominal wall and transplanted to a patient’s face after fracture of the zygomatic arch, with superb results. Lexer was a pioneer in the management of disfigurement injuries in soldiers during World War I. In 1919, he published a two-volume book titled Die freien Transplantationen (“Free Transplantations”), in which he described all the types of fat grafts available at that time.
In the 1930s, surgeons realized that such promising results faded over time, mainly due to unpredictable fat tissue reabsorption, a high propensity for the formation of oily cysts and grafts having a tendency to become hard and fibrotic. These disappointing long-term results made fat grafting an obsolete procedure by the 1950s.3 Decades later, the advent of liposuction in the early 1980s revitalized fat grafting, now that syringe harvesting and injection (micro-lipo-injection) had rendered the harvesting process easier. Despite such advances, the problem of adipose tissue reabsorption remained a significant drawback of this procedure, hindering aesthetic results. In the late 1980s, Abel Chajchir, an Argentinean plastic surgeon, described promising long-term results after fat injection. He recommended careful handling of adipose tissue to decrease the potential risk for adipocyte rupture using normal saline irrigation to eliminate dead cells and grafting adipose tissue into well-vascularized tissue.4
In the 1990s, American plastic surgeon Sydney Coleman recommended harvesting adipose tissue with a 3-mm blunt cannula connected to a 10-mL syringe at low negative pressure to decrease fat cell rupture, centrifugation to separate other fatty tissue components from adipocytes and tunneling the fat using an 18-gauge cannula in close proximity to vascularized tissue.5,6 Cheriyan et al confirmed that low-pressure abdominal lipo-aspiration resulted in a higher rate of cell viability than high-pressure abdominal fat grafting.7 Several studies were conducted demonstrating the presence of adult mesenchymal stem cells and adipose-derived stem cells (ASCs) in subcutaneous tissue and the regenerative properties of ASCs in damaged or missing tissues in human patients.8-10 Fat grafting has progressed considerably over the years and, ultimately, become one of the most popular procedures in the aesthetic and reconstructive fields.11-13
References
- Neuber F. Fettransplantation. Chir Kongr Verhandl Dsch Gesellch Chir. 1893;22:66.
- Czerny V. Plastischer Ersatz der Brustdruse durch ein Lipom. Zentralbl Chir. 1895;27:72.
- Peer LA. Loss of weight and volume in human fat grafts: with postulation of a “cell survival theory.” Plast Reconstr Surg. 1950;5(3):217-230.
- Chajchir A, Benzaquen I. Fat-grafting injection for soft-tissue augmentation. Plast Reconstr Surg. 1989;84(6):921-934.
- Coleman SR. The technique of periorbital lipoinfiltration. Oper Tech Plast Reconstr Surg. 1994;1(3):120-126.
- Coleman SR. Long-Term survival of fat transplants: controlled demonstrations. Aesthetic Plast Surg. 1995;19(5):421-425.
- Cheriyan T, Kao HK, Qiao X, et al. Low harvest pressure enhances autologous fat graft viability. Plast Reconstr Surg. 2014;133(6):1365-1368.
- Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211-228.
- Rodriguez AM, Elabd C, Amri EZ, et al. Thehuman adipose tissue is a source of multipotent stem cells. Biochimie. 2005;87(1):125-128.
- Rigotti G, Marchi A, GaliÈ M, et al. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: a healing process mediated by adipose-derived adult stem cells. Plast Reconstr Surg. 2007;119(5):1409-1422.
- Mazzola RF, Mazzola IC. The fascinating history of fat grafting. J Craniofac Surg. 2013;24(4):1069-1071.
- Mazzola RF, Cantarella G, Torretta S, et al. Autologous fat injection to face and neck: from soft tissue augmentation to regenerative medicine. Acta Otorhinolaryngol Ital. 2011;31(2):59-69.
- Varkarakis G. Fat Injection: From Filling to Regeneration. 2nd Edition. Plast Reconstr Surg. 2018;142(6):1665-1666.
This article is from the December 2022 print issue.





Please log in to post a comment